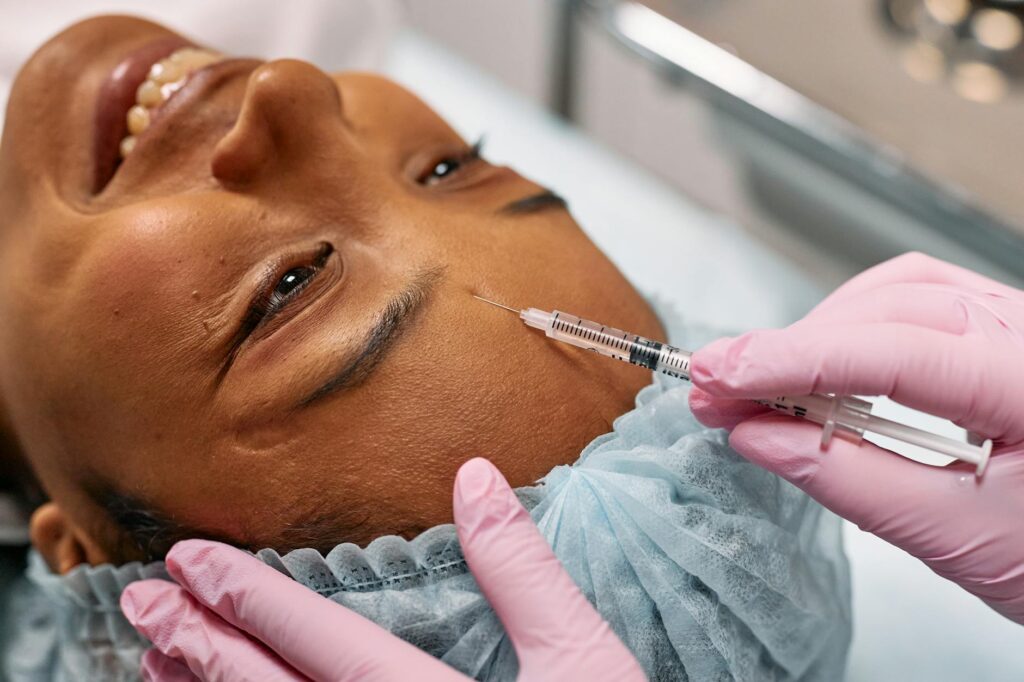
Allergan has announced positive Phase 2 clinical trial results for its investigational next-generation neurotoxin, demonstrating a significantly faster onset of action compared to existing botulinum toxin products. The findings, published in Dermatologic Surgery, showed that patients treated with the novel formulation experienced visible aesthetic improvements within 24 hours, compared to the typical 3-7 day onset period associated with current neurotoxins on the market.
The randomized, double-blind study enrolled 187 participants seeking treatment for moderate to severe glabellar lines. Results indicated that 68% of patients treated with Allergan’s experimental neurotoxin achieved at least a one-grade improvement on the Facial Wrinkle Scale within the first day following injection, while maintaining a safety profile comparable to approved botulinum toxin type A products. The accelerated onset represents a meaningful advancement in aesthetic neurotoxin technology, which has remained relatively unchanged since the approval of Botox Cosmetic over two decades ago.
This development arrives as the aesthetic neurotoxin market continues to expand rapidly, with global revenues projected to exceed $7 billion annually. Faster-acting formulations address a key patient demand for quicker results, particularly among individuals seeking treatments ahead of special events or those with busy schedules who want to minimize the waiting period for visible effects.
If the compound successfully progresses through Phase 3 trials and gains regulatory approval, it could reshape competitive dynamics in the aesthetic injectables space. For patients, a next-generation neurotoxin offering faster results while maintaining established safety standards could provide greater convenience and satisfaction with aesthetic procedures. Allergan has indicated plans to initiate larger-scale Phase 3 trials in the coming months to further evaluate efficacy, duration of effect, and long-term safety outcomes.