The U.S. Food and Drug Administration has granted orphan drug designation to an exosome-based therapy for the treatment of glioblastoma, marking a significant regulatory milestone for this emerging therapeutic approach. The designation, reported by Endpoints News, provides the developer with various incentives including tax credits for clinical trials, waiver of FDA application fees, and seven years of market exclusivity upon approval.
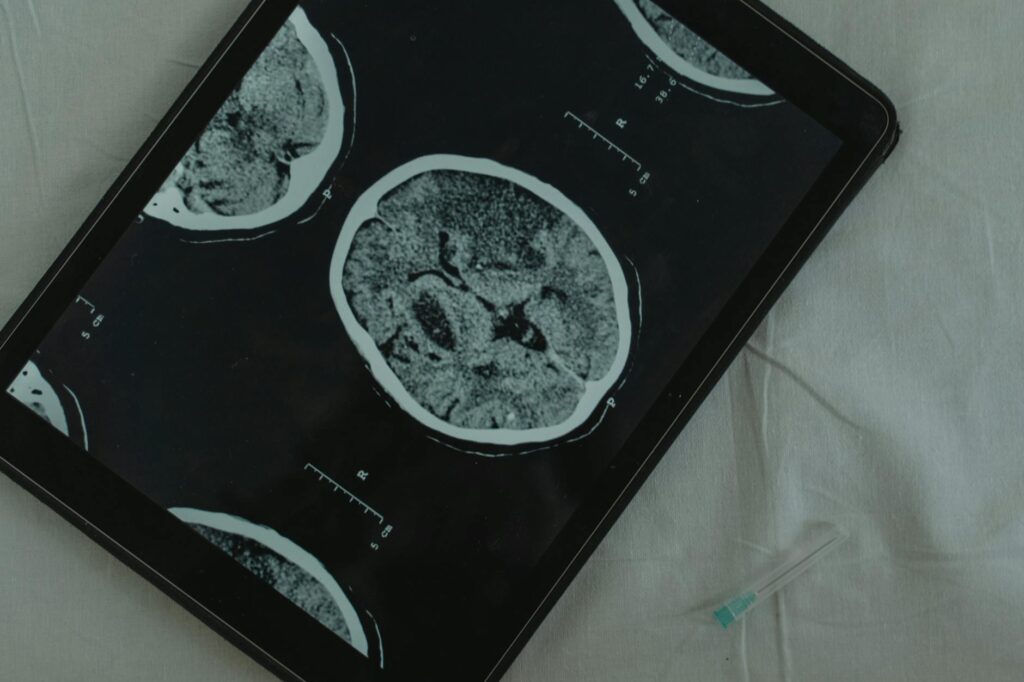
Glioblastoma is the most aggressive and deadly form of brain cancer, with approximately 10,000 new cases diagnosed annually in the United States. Despite standard treatments including surgery, radiation, and chemotherapy, the median survival time remains only 12-18 months after diagnosis. The orphan drug designation applies to diseases affecting fewer than 200,000 people in the U.S., making glioblastoma eligible for this special regulatory pathway.
Exosomes are tiny vesicles naturally produced by cells that can carry therapeutic cargo across the blood-brain barrier, a major obstacle in treating brain cancers. This particular therapy leverages exosomes’ ability to deliver treatment directly to tumor cells while potentially minimizing systemic side effects. The approach represents a novel application of what researchers call “nature’s delivery vehicles” for targeting one of oncology’s most challenging diseases.
The FDA orphan drug designation is not an approval but rather a recognition that the therapy addresses an unmet medical need in a rare disease. It signals regulatory support for continued development while providing financial and procedural advantages that can accelerate the path toward clinical trials and eventual market approval. For glioblastoma patients who face limited treatment options and poor prognosis, the advancement of innovative therapies through regulatory milestones offers a measure of hope in a disease area desperate for breakthroughs.